One of the byproducts of this process is hydrogen ions (H⁺), which can lower the pH in the environment. A decreasing pH has a negative effect on the efficiency of the biological process because many microorganisms involved are sensitive to acidic conditions. Too low a pH can lead to reduced enzyme activity and growth rate of the microbes, slowing or even stopping the reduction process. In particular, the Dehalococcoides mccartyi strains, those responsible for the complete conversion of PCE and TCE to ethylene, are very sensitive. At pH < 6, their activity drops to zero.
Applying pH buffering, such as carbonate buffers or the addition of alkaline substances such as sodium bicarbonate or magnesium hydroxide , prevents the pH from dropping too much. This maintains a stable, neutral to slightly alkaline environment in which the microorganisms involved can function optimally. This makes reductive dechlorination more efficient and allows the remediation process to proceed successfully.
In short, pH buffering is essential to support microbial activity and make the remediation process of CVOCs effective and sustainable.
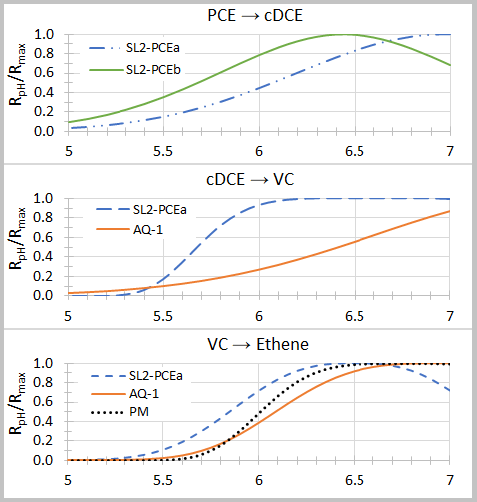
Figure 1. Relative dechlorination rates (RpH /Rmax) versus pH for consortia SL2-PCEa, SL2-PCEb, AQ-1 and PM (adapted from Lacroix et al. 2014)[32]. Source: Enviro.wiki
pH Buffers
QM Environmental International supplies various buffers of different strengths based on calcium carbonate and sodium bicarbonate and magnesium hydroxide. For groundwater with very low pHs, CoBupHMg is an ideal buffer with a very long-lasting effect.


